About
Invivotek, a member of Genesis Biotechnology Group™ (GBG) is a pre-clinical contract research organization specializing in translational studies. We are located in “Einstein’s Alley”, the research and technology corridor in Hamilton, New Jersey, easily accessible to biotech and pharmaceutical companies and academic biomedical centers in the New York / New Jersey / Pennsylvania areas. Invivotek was established in 2012 and launched its first commercial project in 2013. The Invivotek team consists of former Xenogen Biosciences employees, the group that was providing pre-clinical in vivo contract research for over 25 years.
Invivotek offers both custom and standard pre-clinical services to support drug discovery and development programs for our clients. Company’s portfolio includes bioassays designed to support in vivo testing of compounds related to Immunology and Inflammation, Diabetes, Cardiovascular, Obesity, Neurodegenerative disorders, Cancer and other diseases. Invivotek’s in vivo testing capabilities are supported by biochemical and molecular biology techniques as well as functional assays using primary cell cultures established from various mouse tissues under disease model and normal conditions. These assays provide tools to study the mechanism of action of various test therapeutics or potential target genes and to explore drug efficacy biomarkers.
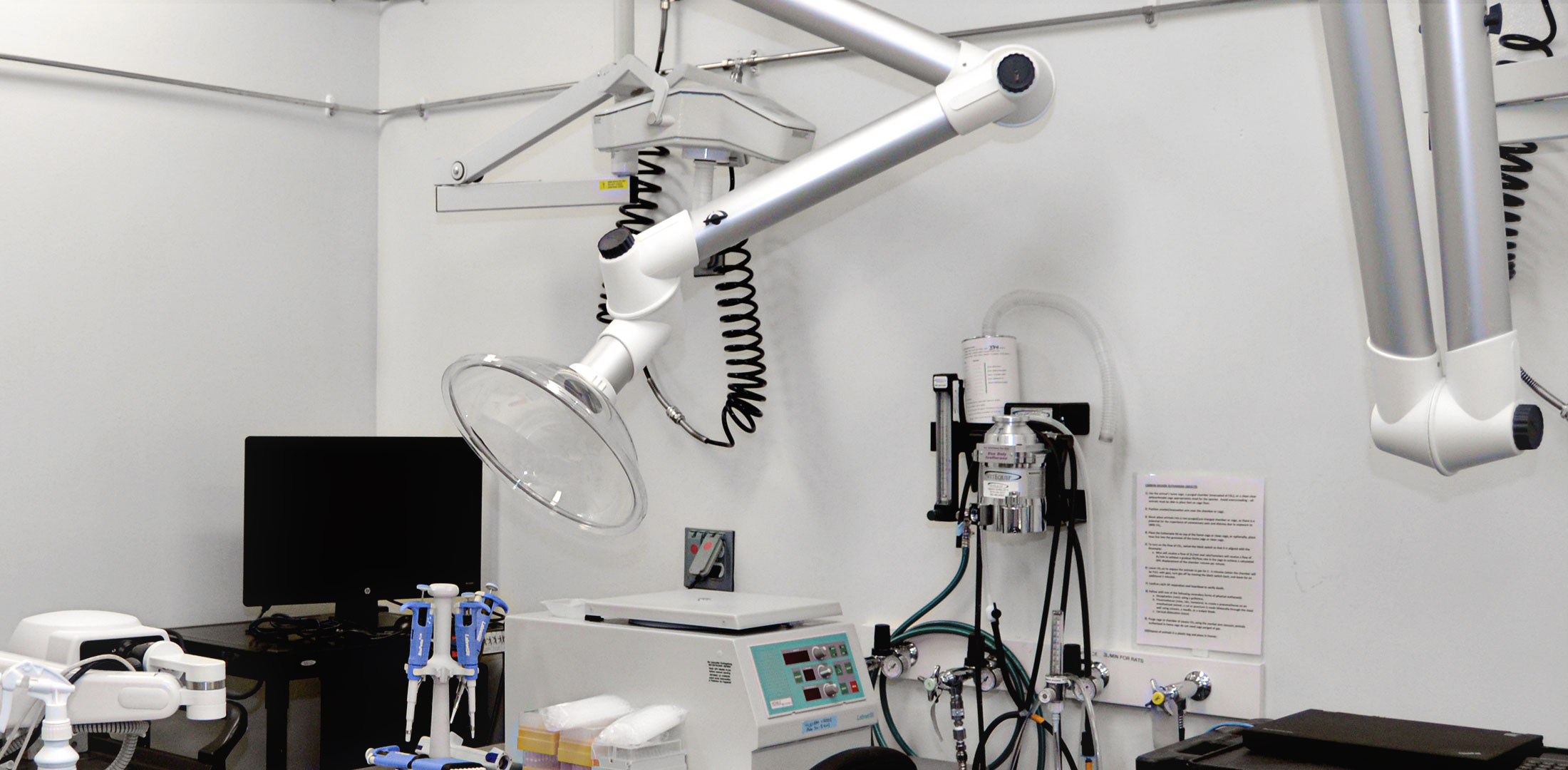
Our vivarium is housed in a new award-winning 19,000 sq. ft. barrier facility that uses the most advanced laboratory technology and precise environmental control. Invivotek’s “clean and dirty corridor” design enables an optimal flow of animals, personnel, equipment and supplies to prevent cross-contaminations.
The collective experience across multiple therapeutic areas and the business and project management skills of the team working in the new state-of-the-art animal facility – all positions Invivotek to become a leading provider of preclinical in vivo services.